UPOs pushing boundaries in terpene chemistry
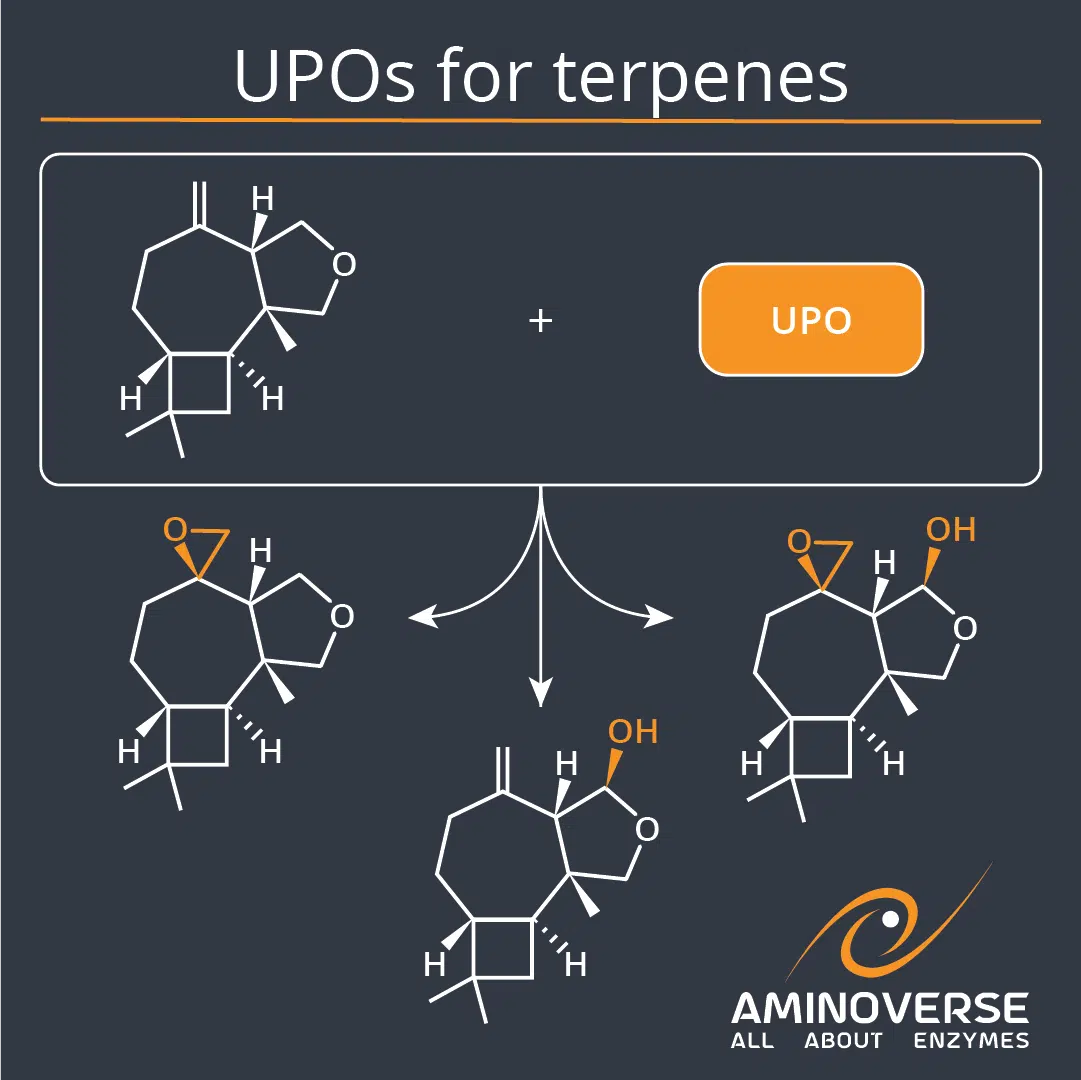
The team of Henry Struwe, Christopher Grimm, Gerald Dräger, Sascha Beutel, Miguel Alcalde, Andreas Kirschning and Selin Kara explored how UPOs act on non-natural terpenes. Screening of UPOs against a synthetic tricyclic oxaterpenoid derived from a modified farnesyl pyrophosphate ether resulted in many novel products. Two Aminoverse UPOs performed at semi-preparative scale, enabling isolation of compounds derived from epoxidation and selective C–H activation.
In pharma and flavours & fragrances, access to oxidised terpenoid analogues has often depended on complex chemical routes. UPOs accept unnatural substrates and deliver transformations that are difficult or inefficient to achieve non-enzymatically. This opens paths to new molecule space with improved efficiency, selectivity and sustainability.
The Aminoverse panel offers a major advantage by providing many active UPOs ready for screening. That accelerates discovery of enzymes matched to challenging substrates, and reduces time to scale for promising new oxidised terpenoid-based products.
Expanding the “Terpenome”: Applications of Unspecific Peroxygenases (UPOs) in Oxidations of Unnatural Terpenoids
“An unnatural tricyclic oxaterpenoid, obtained by treatment of the sesquiterpene synthase presilphiperfolan-8β-ol synthase (BcBOT2) with an unnatural farnesyl pyrophosphate ether derivative, itself obtained by chemical synthesis, was converted in oxidation studies as part of a broad screening program by selected unspecific peroxygenases (UPOs). Product analysis revealed that Agrocybe aegerita UPO, its mutant PaDa-I, and two commercial ones, UPO54 and UPO49, provided new oxidation products with sufficient efficiency for subsequent upscaling that allowed product isolation and structure elucidation. As such new terpene-based oxiranes and hemiacetals were formed by UPO-mediated epoxidations and CH-activation. The structure elucidation was further supported by comparison with products generated by chemical oxidation.”
